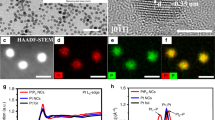
Abstract
Two-electron oxygen reduction offers a route to H2O2 that is potentially cost-effective and less energy-intensive than the industrial anthraquinone process. However, the catalytic performance of the highest performing prior heterogeneous electrocatalysts to H2O2 has lain well below the >300 mA cm−2 needed for capital efficiency. Herein, guided by computation, we present a supramolecular approach that utilizes oxygen functional groups in a carbon nanotube substrate that—when coupled with a cobalt phthalocyanine catalyst—improve cobalt phthalocyanine adsorption, preventing agglomeration; and that further generate an electron-deficient Co centre whose interaction with the key H2O2 intermediate is tuned towards optimality. The catalysts exhibit an overpotential of 280 mV at 300 mA cm−2 with turnover frequencies over 50 s−1 in a neutral medium, an order of magnitude higher activity compared with the highest performing prior H2O2 electrocatalysts. This performance is sustained for over 100 h of operation.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data supporting the findings of this study are provided with the paper and its Supplementary Information files. Geometries are also available at https://github.com/hitarth64/CoPc-CNT. Further requests about data can be addressed to the corresponding authors. Source data are provided with this paper.
References
Hydrogen Peroxide (HP): 2015 World Market Outlook (Merchant Research and Consulting, 2015).
Siahrostami, S. et al. A review on challenges and successes in atomic-scale design of catalysts for electrochemical synthesis of hydrogen peroxide. ACS Catal. 10, 7495–7511 (2020).
Perry, S. C. et al. Electrochemical synthesis of hydrogen peroxide from water and oxygen. Nat. Rev. Chem. 3, 442–458 (2019).
Jung, E. at al. Atomic-level tuning of Co-N-C catalyst for high-performance electrochemical H2O2 production. Nat. Mater. 19, 436–442 (2020).
Jung, E., Shin, H., Hooch Antink, W., Sung, Y. E. & Hyeon, T. Recent advances in electrochemical oxygen reduction to H2O2: Catalyst and cell design. ACS Energy Lett. 5, 1881–1892 (2020).
Zhang, X. L. et al. Strongly coupled cobalt diselenide monolayers for selective electrocatalytic oxygen reduction to H2O2 under acidic conditions. Angew. Chem. Int. Ed. 60, 2–11 (2021).
Xia, C., Xia, Y., Zhu, P., Fan, L. & Wang, H. Direct electrosynthesis of pure aqueous H2O2 solutions up to 20% by weight using a solid electrolyte. Science 366, 226–231 (2019).
Han, Z., Horak, K. T., Lee, H. B. & Agapie, T. Tetranuclear Manganese Models of the OEC displaying hydrogen bonding interactions: Application to electrocatalytic water oxidation to hydrogen peroxide. J. Am. Chem. Soc. 139, 9108–9111 (2017).
Costentin, C., Drouet, S., Robert, M. & Saveant, J. M. A local proton source enhances CO2 electroreduction to CO by a molecular Fe catalyst. Science 338, 90–94 (2012).
Buss, J. A. & Agapie, T. Four-electron deoxygenative reductive coupling of carbon monoxide at a single metal site. Nature 529, 72–75 (2016).
Liu, C. et al. Intrinsic activity of metal metal centers in metal-nitrogen-carbon single-atom catalysts for hydrogen peroxide synthesis. J. Am. Chem. Soc. 142, 21861–21871 (2020).
Wang, Y. H., Pegis, M. L., Mayer, J. M. & Stahl, S. S. Molecular cobalt catalyst for O2 reduction: Low-overpotential production of H2O2 and comparison with Iron-based catalysts. J. Am. Chem. Soc. 139, 16458–16461 (2017).
Honda, T., Kojima, T. & Fukuzumi, S. Proton-coupled electron-transfer reduction of dioxygen catalyzed by a saddle-distorted cobalt phthalocyanine. J. Am. Chem. Soc. 134, 4196–4206 (2012).
Wu, Y., Jiang, Z., Lu, X., Liang, Y. & Wang, H. Domino electroreduction of CO2 to methanol on a molecular catalyst. Nature 575, 639–642 (2019).
Severy, L. et al. Immobilization of molecular catalysts on electrode surfaces using host-guest interactions. Nat. Chem. 13, 523–529 (2021).
Zhu, M., Ye, R., Jin, K., Lazouski, N. & Manthiram, K. Elucidating the reactivity and mechanism of CO2 electroreduction at highly dispersed cobalt phthalocyanine. ACS Energy Lett. 3, 1381–1386 (2018).
Wang, H. et al. Direct and continuous strain control of catalysts with tunable battery electrode materials. Science 354, 1031–1036 (2016).
He, T. et al. Mastering the surface strain of platinum catalysts for efficient electrocatalysis. Nature 598, 76–81 (2021).
Luo, M. et al. PdMo bimetallene for oxygen reduction catalysis. Nature 574, 81–85 (2019).
Bok, J. et al. Designing atomically dispersed Au on tensile-strained Pd for efficient CO2 electroreduction to formate. J. Am. Chem. Soc. 143, 5386–6395 (2021).
Siahrostami, S. et al. Enabling direct H2O2 production through rational electrocatalyst design. Nat. Mater. 12, 1137–1143 (2013).
Escudero-Escribano, M. et al. Tuning the activity of Pt alloy electrocatalysts by means of the lanthanide contraction. Science 352, 73–76 (2016).
Chen, C. et al. Highly crystalline multimetallic nanoframes with three-dimensional electrocatalytic surfaces. Science 343, 1339–1343 (2014).
Zhang, B. et al. Homogeneously dispersed multimetal oxygen-evolving catalysts. Science 352, 333–337 (2016).
Lu, Z. et al. High-efficiency oxygen reduction to hydrogen peroxide catalysed by oxidized carbon materials. Nat. Catal. 1, 156–162 (2018).
Guo, D. et al. Active sites of nitrogen-doped carbon materials for oxygen reduction reaction clarified using model catalysts. Science 351, 361–365 (2016).
Jiao, Y., Zheng, Y., Jaroniec, M. & Qiao, S. Z. Origin of the electrocatalytic oxygen reduction activity of graphene-based catalysts: A roadmap to achieve the best performance. J. Am. Chem. Soc. 136, 4394–4403 (2014).
Gu, J., Hsu, C. S., Bai, L., Chen, H. M. & Hu, X. Atomically dispersed Fe3+ sites catalyze efficient CO2 electroreduction to CO. Science 364, 1091–1094 (2019).
Seh, Z. W. et al. Combining theory and experiment in electrocatalysis: Insights into materials design. Science 355, eaad4998 (2017).
Chen, S. et al. Chemical identification of catalytically active sites on oxygen-doped carbon nanosheet to decipher the high activity for electro-synthesis hydrogen peroxide. Angew. Chem. Int. Ed. 60, 16607–16614 (2021).
Cao, P. et al. Durable and selective electrochemical H2O2 synthesis under a large current enabled by the cathode with highly hydrophobic three-phase architecture. ACS Catal. 11, 13797–13808 (2021).
Zhao, Q. et al. Approaching a high-rate and sustainable production of hydrogen peroxide: oxygen reduction on Co-N-C single-atom electrocatalysts in simulated seawater. Energy Environ. Sci. 14, 5444–5456 (2021).
Pan, Y. et al. Design of single-atom Co-N5 catalytic site: A robust electrocatalyst for CO2 reduction with nearly 100% CO selectivity and remarkable stability. J. Am. Chem. Soc. 140, 4218–4221 (2018).
Ren, S. et al. Molecular electrocatalysts can mediate fast, selective CO2 reduction in a flow cell. Science 365, 367–369 (2019).
Zhang, X. et al. Molecular engineering of dispersed nickel phthalocyanines on carbon nanotubes for selective CO2 reduction. Nat. Energy 5, 684–692 (2020).
Schild, J. et al. Approaching industrially relevant current densities for hydrogen oxidation with a bioinspired molecular catalytic material. J. Am. Chem. Soc. 143, 18150–18158 (2021).
Jin, H. et al. Unprecedentedly high activity and selectivity for hydrogenation of nitroarenes with single atomic Co1-N3P1 sites. Nat. Commun. 13, 723 (2022).
Mun, Y. et al. Versatile strategy for tuning ORR activity of a single fe-n4 site by controlling electron-withdrawing/donating properties of a carbon plane. J. Am. Chem. Soc. 141, 6254–6262 (2019).
Vijay, S. et al. Unified mechanistic understanding of CO2 reduction to CO on transition metal and single atom catalysts. Nat. Catal. 4, 1024–1031 (2021).
Ramaswamy, N. & Mukerjee, S. Influence of inner- and outer-sphere electron transfer mechanisms during electrocatalysis of oxygen reduction in alkaline media. J. Phys. Chem. C. 115, 18015–18026 (2011).
Blizanac, B. B., Ross, P. N. & Markovic, N. M. Oxygen electroreduction on Ag(1 1 1): The pH effect. Electrochim. Acta 52, 2264–2271 (2007).
Luna, P. D. et al. What would it take for renewably powered electrosynthesis to displace petrochemical processes? Science 364, eaav3506 (2019).
Yu, X. et al. Boron-doped graphene for electrocatalytic N2 reduction. Joule 2, 1610–1622 (2018).
Wang, M. et al. CO2 electrochemical catalytic reduction with a highly active cobalt phthalocyanine. Nat. Commun. 10, 3602 (2019).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953–17979 (1994).
Wang, L., Maxisch, T. & Ceder, G. Oxidation energies of transition metal oxides within the GGA+U framework. Phys. Rev. B 73, 195107 (2006).
Tkatchenko, A., DiStasio, R. A. Jr., Car, R. & Scheffler, M. Accurate and Efficient Method for Many-Body van der Waals Interactions. Phys. Rev. Lett. 108, 236402 (2012).
Man, I. C. et al. Universality in oxygen evolution electrocatalysis on oxide surfaces. ChemCatChem 3, 1159–1165 (2011).
Nørskov, J. K. et al. Origin of the overpotential for oxygen reduction at a fuel-cell cathode. J. Phys. Chem. B 108, 17886–17892 (2004).
Mathew, K. et al. Implicit solvation model for density-functional study of nanocrystal surfaces and reaction pathways. J. Chem. Phys. 140, 084106 (2014).
Mathew, K. et al. Implicit self-consistent electrolyte model in plane-wave density-functional theory. J. Chem. Phys. 151, 234101 (2019).
Acknowledgements
This work was supported by the Natural Gas Innovation Fund, the Natural Sciences and Engineering Research Council of Canada, the Natural Resources Canada Clean Growth Program, and the Ontario Research Fund—Research Excellence programme. All DFT computations were performed on the Niagara supercomputer at the SciNet HPC Consortium. SciNet is funded by the Canada Foundation for Innovation, the Government of Ontario, the Ontario Research Fund Research Excellence Program, and the University of Toronto. This work was also supported by the Research Center Program of the IBS (IBS-R006-A2, Y.-E.S; IBS-R006-D1, T.H) in Korea.
Author information
Authors and Affiliations
Contributions
E.H.S., Y.-E.S. and T.H. supervised the project. B.-H.L. conceived the idea. B.-H.L., H.S., A.S.R. and H.C. designed and performed the experiments. H.C. with the help of P.O. carried out DFT calculations. R.D., I.G., G.L., S.P., H.S.L., E.S., J.W., J.Z., Z.C. and Y.C. contributed on material synthesis, characterization and electrochemical measurements. R.K.M. and D.S. assisted with electrochemical system design. B.-H.L., H.S., A.S.R., H.C., T.H., Y.-E.S. and E.H.S. wrote the manuscript. All authors commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Catalysis thanks Hailiang Wang and the other, anonymous, reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary figs. 1–37, notes 1–4 and tables 1–3.
Supplementary Data 1
Structural data.
Source data
Source Data Fig. 1
Source data fig. 1.
Source Data Fig. 3
Source data fig. 3.
Source Data Fig. 4
Source data fig. 4.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lee, BH., Shin, H., Rasouli, A.S. et al. Supramolecular tuning of supported metal phthalocyanine catalysts for hydrogen peroxide electrosynthesis. Nat Catal 6, 234–243 (2023). https://doi.org/10.1038/s41929-023-00924-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-023-00924-5