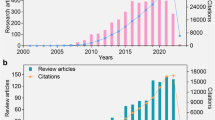
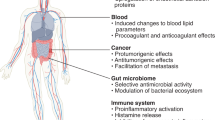
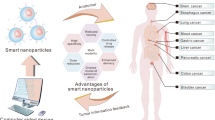
Abstract
Oxide nanoparticles have garnered significant attention in biomedical research owing to the numerous available synthetic approaches and highly tunable physicochemical properties, which enable diverse functions within biological systems. These nanoparticles can be broadly categorized based on their characteristics useful for biomedical applications. Magnetic oxide nanoparticles, for instance, are prominently used as contrast agents in MRI and as mediators to generate heat, mechanical force or electricity for therapy. Catalytic oxide nanoparticles can generate or eliminate reactive oxygen species, which are central to numerous biological processes. Porous oxide nanoparticles are adept at loading dye or drug molecules, making them invaluable for bioimaging and therapeutic interventions. In this Review, we highlight strategies for the fabrication and advanced engineering of oxide nanoparticles tailored for biomedical applications. We primarily focus on iron oxide, ceria and silica nanoparticles, delving into their diagnostic and therapeutic potentials. We also discuss future prospects and the challenges that must be addressed to meet clinical needs.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Murray, C. B., Norris, D. J. & Bawendi, M. G. Synthesis and characterization of nearly monodisperse CdE (E = sulfur, selenium, tellurium) semiconductor nanocrystallites. J. Am. Chem. Soc. 115, 8706–8715 (1993).
Alivisatos, A. P. Semiconductor clusters, nanocrystals, and quantum dots. Science 271, 933–937 (1996).
Sun, S., Murray, C. B., Weller, D., Folks, L. & Moser, A. Monodisperse FePt nanoparticles and ferromagnetic FePt nanocrystal superlattices. Science 287, 1989–1992 (2000).
Elghhanian, R. et al. Selective colorimetric detection of polynucleotides based on the distance-dependent optical properties of gold nanoparticles. Science 277, 1078–1081 (1997).
Bruchez, M. Jr. et al. Semiconductor nanocrystals as fluorescent biological labels. Science 281, 2013–2016 (1998).
Chan, W. C. W. & Nie, S. Quantum dot bioconjugates for ultrasensitive nonisotopic detection. Science 281, 2016–2018 (1998).
Huang, H., Feng, W., Chen, Y. & Shi, J. Inorganic nanoparticles in clinical trials and translations. Nano Today 35, 100972 (2020).
Geng, H. et al. Noble metal nanoparticle biosensors: from fundamental studies toward point-of-care diagnostics. Acc. Chem. Res. 55, 593–604 (2022).
Hirschbiegel, C.-M. et al. Inorganic nanoparticles as scaffolds for bioorthogonal catalysts. Adv. Drug Deliv. Rev. 195, 114730 (2023).
Pelaz, B. et al. Diverse applications of nanomedicine. ACS Nano 11, 2313–2381 (2017).
Kwon, H. J. et al. Large-scale synthesis and medical applications of uniform sized metal oxide nanoparticles. Adv. Mater. 30, 1704290 (2018).
Park, J. et al. Ultra-large-scale synthesis of monodisperse nanocrystals. Nat. Mater. 3, 891–895 (2004).
Djurišić, A. B. et al. Toxicity of metal oxide nanoparticles: mechanisms, characterization, and avoiding experimental artefacts. Small 11, 26–64 (2015).
Jun, Y.-W. et al. Nanoscale size effect of magnetic nanocrystals and their utilization for cancer diagnosis via magnetic resonance imaging. J. Am. Chem. Soc. 127, 5732–5733 (2005).
Shin, T.-H. et al. High-resolution T1 MRI via renally clearable dextran nanoparticles with an iron oxide shell. Nat. Biomed. Eng. 5, 252–263 (2021).
Lee, N. et al. Iron oxide based nanoparticles for multimodal imaging and magnetoresponsive therapy. Chem. Rev. 115, 10637–10689 (2015).
Ho, D., Sun, X. & Sun, S. Monodisperse magnetic nanoparticles for theranostic applications. Acc. Chem. Res. 44, 875–882 (2011).
Ma, Z., Mohapatra, J., Wei, K., Liu, J. P. & Sun, S. Magnetic nanoparticles: synthesis, anisotropy, and applications. Chem. Rev. 123, 3904–3943 (2021).
Scott, J. F. Applications of magnetoelectrics. J. Mater. Chem. 22, 4567–4574 (2012).
Carneiro, J. S., Williams, J., Gryko, A., Herrera, L. P. & Nikolla, E. Embracing the complexity of catalytic structures: a viewpoint on the synthesis of nonstoichiometric mixed oxides for catalysis. ACS Catal. 10, 516–527 (2020).
Tu, Z. et al. Design of therapeutic biomaterials to control inflammation. Nat. Rev. Mater. 7, 557–574 (2022).
Aragon-Sanabria, V. et al. Ultrasmall nanoparticle delivery of doxorubicin improves therapeutic index for high-grade glioma. Clin. Cancer Res. 28, 2938–2952 (2022).
Kim, D., Shin, K., Kwon, S. G. & Hyeon, T. Synthesis and biomedical applications of multifunctional nanoparticles. Adv. Mater. 30, 1802309 (2018).
Bee, A., Massart, R. & Neveu, S. Synthesis of very fine maghemite particles. J. Magn. Magn. Mater. 149, 6–9 (1995).
Stöber, W., Fink, A. & Bohn, E. Controlled growth of monodisperse silica spheres in the micron size range. J. Colloid Interface Sci. 26, 62–69 (1968).
Peng, X., Wickham, J. & Alivisatos, A. P. Kinetics of II–VI and III–V colloidal semiconductor nanocrystal growth: ‘focusing’ of size distributions. J. Am. Chem. Soc. 120, 5343–5344 (1998).
Peng, X. et al. Shape control of CdSe nanocrystals. Nature 404, 59–61 (2000).
Boles, M. A., Ling, D., Hyeon, T. & Talapin, D. V. The surface science of nanocrystals. Nat. Mater. 15, 141–153 (2016).
Silvera Batista, C. A., Larson, R. G. & Kotov, N. A. Nonadditivity of nanoparticle interactions. Science 350, 1242477 (2015).
Baumgartner, J. et al. Nucleation and growth of magnetite from solution. Nat. Mater. 12, 310–314 (2013).
Lee, J., Yang, J., Kwon, S. G. & Hyeon, T. Nonclassical nucleation and growth of inorganic nanoparticles. Nat. Rev. Mater. 1, 16034 (2016).
Li, D. et al. Nanoparticle assembly and oriented attachment: correlating controlling factors to the resulting structures. Chem. Rev. 123, 3127–3159 (2023).
Kwon, S. G. et al. Heterogeneous nucleation and shape transformation of multicomponent metallic nanostructures. Nat. Mater. 14, 215–223 (2015).
Wheeler, K. E. et al. Environmental dimensions of the protein corona. Nat. Nanotechnol. 16, 617–629 (2021).
Stater, E. P., Sonay, A. Y., Hart, C. & Grimm, J. The ancillary effects of nanoparticles and their implications for nanomedicine. Nat. Nanotechnol. 16, 1180–1194 (2021).
Karakoti, A. S., Das, S., Thevuthasan, S. & Seal, S. Pegylated inorganic nanoparticles. Angew. Chem. Int. Ed. 50, 1980–1994 (2011).
Pelaz, B. et al. Surface functionalization of nanoparticles with polyethylene glycol: effects on protein adsorption and cellular uptake. ACS Nano 9, 6996–7008 (2015).
Fu, J., Wu, E., Li, G., Wang, B. & Zhan, C. Anti-PEG antibodies: current situation and countermeasures. Nano Today 55, 102163 (2024).
Poon, W., Kingston, B. R., Ouyang, B., Ngo, W. & Chan, W. C. W. A framework for designing delivery systems. Nat. Nanotechnol. 15, 819–829 (2020).
Dilliard, S. A. & Siegwart, D. J. Passive, active and endogenous organ-targeted lipid and polymer nanoparticles for delivery of genetic drugs. Nat. Rev. Mater. 8, 282–300 (2023).
Nguyen, L. N. M. et al. The mechanisms of nanoparticle delivery to solid tumours. Nat. Rev. Bioeng. 2, 201–213 (2024).
Mahmoudi, M., Landry, M. P., Moore, A. & Coreas, R. The protein corona from nanomedicine to environmental science. Nat. Rev. Mater. 8, 422–438 (2023).
Dillard, S. A., Cheng, Q. & Siegwart, D. J. On the mechanism of tissue-specific mRNA delivery by selective organ targeting nanoparticle. Proc. Natl Acad. Sci. USA 118, e2109256118 (2021).
Ngo, W. et al. Identifying cell receptors for the nanoparticle protein corona using genome screens. Nat. Chem. Biol. 18, 1023–1031 (2022).
Choi, H. S. et al. Renal clearance of quantum dots. Nat. Biotechnol. 25, 1165–1170 (2007).
van Leent, M. M. T. et al. Regulating trained immunity with nanomedicine. Nat. Rev. Mater. 7, 465–481 (2022).
Poon, W. et al. Elimination pathways of nanoparticles. ACS Nano 13, 5785–5798 (2019).
Burns, A. A. et al. Fluorescent silica nanoparticles with efficient urinary excretion for nanomedicine. Nano Lett. 9, 442–448 (2009).
Bourquin, J. et al. Biodistribution, clearance, and long-term fate of clinically relevant nanomaterials. Adv. Mater. 30, 1704307 (2018).
Mahmoudi, M., Hofmann, H., Rothen-Rutishauser, B. & Petri-Fink, A. Assessing the in vitro and in vivo toxicity of superparamagnetic iron oxide nanoparticles. Chem. Rev. 112, 2323–2338 (2012).
Arami, H., Khandhar, A., Liggitt, D. & Krishnan, K. M. In vivo delivery, pharmacokinetics, biodistribution and toxicity of iron oxide nanoparticles. Chem. Soc. Rev. 44, 8576–8607 (2015).
Feliu, N. et al. In vivo degeneration and the fate of inorganic nanoparticles. Chem. Soc. Rev. 45, 2440–2457 (2016).
Mahmoudi, M., Laurent, S., Shokrgozar, M. A. & Hosseinkhani, M. Toxicity evaluations of superparamagnetic iron oxide nanoparticles: cell ‘vision’ versus physicochemical properties of nanoparticles. ACS Nano 5, 7263–7276 (2011).
World Health Organisation & International Agency for Research on Cancer. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans: Silica, Some Silicates, Coal Dust and Para-Aramid Fibrils Vol. 68 (IARC, 1997).
US Food and Drug Administration. GRAS Substances (SCOGS) database. FDA https://www.fda.gov/food/generally-recognized-safe-gras/gras-substances-scogs-database (2020).
Croissant, J. G., Butler, K. S., Zink, J. I. & Brinker, C. J. Synthetic amorphous silica nanoparticles: toxicity, biomedical and environmental implications. Nat. Rev. Mater. 5, 886–909 (2020).
Zhang, H. et al. Processing pathway dependence of amorphous silica nanoparticle toxicity: colloidal vs pyrolytic. J. Am. Chem. Soc. 134, 15790–15804 (2012).
Chen, Y. et al. Colloidal HPMO nanoparticles: silica-etching chemistry tailoring, topological transformation, and nano-biomedical applications. Adv. Mater. 25, 3100–3105 (2013).
Slowing, I. I., Wu, C.-W., Vivero-Escoto, J. L. & Lin, V. S.-Y. Mesoporous silica nanoparticles for reducing hemolytic activity towards mammalian red blood cells. Small 5, 57–62 (2009).
Kim, B. H. et al. Large-scale synthesis of uniform and extremely small-sized iron oxide nanoparticles for high-resolution T1 magnetic resonance imaging contrast agents. J. Am. Chem. Soc. 133, 12624–12631 (2011).
Lu, Y. et al. Iron oxide nanoclusters for T1 magnetic resonance imaging of non-human primates. Nat. Biomed. Eng. 1, 637–643 (2017).
Li, H. & Meade, T. J. Molecular magnetic resonance imaging with Gd(III)-based contrast agents: challenges and key advances. J. Am. Chem. Soc. 141, 17025–17041 (2019).
Ning, Y. et al. Molecular MRI quantification of extracellular aldehyde pairs for early detection of liver fibrogenesis and response to treatment. Sci. Transl Med. 14, eabq6297 (2022).
Chen, H. H. et al. A nanoparticle probe for the imaging of autophagic flux in live mice via magnetic resonance and near-infrared fluorescence. Nat. Biomed. Eng. 6, 1045–1056 (2022).
Lu, C. et al. Engineering of magnetic nanoparticles as magnetic particle imaging tracers. Chem. Soc. Rev. 50, 8102–8146 (2021).
Yang, X. et al. Nanotechnology enables novel modalities for neuromodulation. Adv. Mater. 33, 2103208 (2021).
Zhou, H., Mayorga-Martinez, C. C., Pané, S., Zhang, L. & Pumera, M. Magnetically driven micro and nanorobots. Chem. Rev. 121, 4999–5041 (2021).
Qiao, R. et al. Magnetic iron oxide nanoparticles for brain imaging and drug delivery. Adv. Drug Deliv. Rev. 197, 114822 (2023).
Kim, D. et al. Synthesis of uniform ferrimagnetic magnetite nanocubes. J. Am. Chem. Soc. 131, 454–455 (2009).
Vreeland, E. C. et al. Enhanced nanoparticle size control by extending LaMer’s mechanism. Chem. Mater. 27, 6059–6066 (2015).
Zhang, H. et al. A hepatocyte-targeting nanoparticle for enhanced hepatobiliary magnetic resonance imaging. Nat. Biomed. Eng. 7, 221–235 (2023).
Lee, N. & Hyeon, T. Designed synthesis of uniformly sized iron oxide nanoparticles for efficient magnetic resonance imaging contrast agents. Chem. Soc. Rev. 41, 2575–2589 (2012).
Min, C. et al. Mechanism of magnetic relaxation switching sensing. ACS Nano 6, 6821–6828 (2012).
Josephson, L., Perez, J. M. & Weissleder, R. Magnetic nanosensors for the detection of oligonucleotide sequences. Angew. Chem. Int. Ed. 40, 3204–3206 (2001).
Perez, J. M., Josephson, L., O’Loughlin, T., Högemann, D. & Weissleder, R. Magnetic relaxation switches capable of sensing molecular interactions. Nat. Biotechnol. 20, 816–820 (2002).
Lu, J. et al. Highly sensitive diagnosis of small hepatocellular carcinoma using pH-responsive iron oxide nanocluster assemblies. J. Am. Chem. Soc. 140, 10071–10074 (2018).
Zhang, P. et al. Quantitative mapping of glutathione within intracranial tumors through interlocked MRI signals of a responsive nanoprobe. Angew. Chem. Int. Ed. 60, 8130–8138 (2021).
Gallo, J. et al. CXCR4-targeted and MMP-responsive iron oxide nanoparticles for enhanced magnetic resonance imaging. Angew. Chem. Int. Ed. 53, 9550–9554 (2014).
Okada, S. et al. Calcium-dependent molecular fMRI using a magnetic nanosensor. Nat. Nanotechnol. 13, 473–477 (2018).
Hsieh, V. et al. Neurotransmitter-responsive nanosensors for T2-weighted magnetic resonance imaging. J. Am. Chem. Soc. 141, 15751–15754 (2019).
Lee, H., Sun, E., Ham, D. & Weissleder, R. Chip–NMR biosensor for detection and molecular analysis of cells. Nat. Med. 14, 869–874 (2008).
Choi, J.-S. et al. Distance-dependent magnetic resonance tuning as a versatile MRI sensing platform for biological targets. Nat. Mater. 16, 537–542 (2017).
Wang, Z. et al. Two-way magnetic resonance tuning and enhanced subtraction imaging for non-invasive and quantitative biological imaging. Nat. Nanotechnol. 15, 482–490 (2020).
Lu, H. et al. A pH-responsive T1–T2 dual-modal MRI contrast agent for cancer imaging. Nat. Commun. 13, 798 (2022).
Wang, C. et al. An electric-field-responsive paramagnetic contrast agent enhances the visualization of epileptic foci in mouse models of drug-resistant epilepsy. Nat. Biomed. Eng. 5, 278–289 (2021).
Subbiah, V. The next generation of evidence-based medicine. Nat. Med. 29, 49–58 (2023).
Xie, X. et al. Magnetic particle imaging: from tracer design to biomedical applications in vasculature abnormality. Adv. Mater. 36, 2306450 (2024).
Tong, S., Quinto, C. A., Zhang, L., Mohindra, P. & Bao, G. Size-dependent heating of magnetic iron oxide nanoparticles. ACS Nano 11, 6808–6816 (2017).
Chen, R., Christiansen, M. G. & Anikeeva, P. Maximizing hysteretic losses in magnetic ferrite nanoparticles via model-driven synthesis and materials optimization. ACS Nano 7, 8990–9000 (2013).
Kang, T. et al. Penetrative and sustained drug delivery using injectable hydrogel nanocomposites for post-surgical brain tumor treatment. ACS Nano 17, 5435–5447 (2023).
Lee, J.-H. et al. Exchange-coupled magnetic nanoparticles for efficient heat induction. Nat. Nanotechnol. 6, 418–422 (2011).
Yun, H. et al. Size- and composition-dependent radio frequency magnetic permeability of iron oxide nanocrystals. ACS Nano 8, 12323–12337 (2014).
Gavilán, H. et al. Magnetic nanoparticles and clusters for magnetic hyperthermia: optimizing their heat performance and developing combinatorial therapies to tackle cancer. Chem. Soc. Rev. 50, 11614–11667 (2021).
Benfenati, F. & Lanzani, G. Clinical translation of nanoparticles for neural stimulation. Nat. Rev. Mater. 6, 1–4 (2021).
Basbaum, A. I., Bautista, D. M., Scherrer, G. & Julius, D. Cellular and molecular mechanisms of pain. Cell 139, 267–284 (2009).
Koivisto, A.-P., Belvisi, M. G., Gaudet, R. & Szallasi, A. Advances in TRP channel drug discovery: from target validation to clinical studies. Nat. Rev. Drug Discov. 21, 41–59 (2022).
Huang, H., Delikanli, S., Zeng, H., Ferkey, D. M. & Pralle, A. Remote control of ion channels and neurons through magnetic-field heating of nanoparticles. Nat. Nanotechnol. 5, 602–606 (2010).
Chen, R., Romero, G., Christiansen, M. G., Mohr, A. & Anikeeva, P. Wireless magnetothermal deep brain stimulation. Science 347, 1477–1480 (2015).
Deisseroth, K. Optogenetics. Nat. Methods 8, 26–29 (2011).
Sebesta, C. et al. Subsecond multichannel magnetic control of select neural circuits in freely moving flies. Nat. Mater. 21, 951–958 (2022).
Seynhaeve, A. L. B., Amin, M., Haemmerich, D., van Rhoon, G. C. & ten Hagen, T. L. M. Hyperthermia and smart drug delivery systems for solid tumor therapy. Adv. Drug Deliv. Rev. 163–164, 125–144 (2020).
Rao, S. et al. Remotely controlled chemomagnetic modulation of targeted neural circuits. Nat. Nanotechnol. 14, 967–973 (2019).
Croissant, J. & Zink, J. I. Nanovalve-controlled cargo release activated by plasmonic heating. J. Am. Chem. Soc. 134, 7628–7631 (2012).
Derfus, A. M. et al. Remotely triggered release from magnetic nanoparticles. Adv. Mater. 19, 3932–3936 (2007).
Kunz, P. C. et al. Metal carbonyls supported on iron oxide nanoparticles to trigger the CO-gasotransmitter release by magnetic heating. Chem. Commun. 49, 4896–4898 (2013).
Riedinger, A. et al. Subnanometer local temperature probing and remotely controlled drug release based on azo-functionalized iron oxide nanoparticles. Nano Lett. 13, 2399–2406 (2013).
Liu, S. et al. Treatment of infarcted heart tissue via the capture and local delivery of circulating exosomes through antibody-conjugated magnetic nanoparticles. Nat. Biomed. Eng. 4, 1063–1075 (2020).
Mirvakili, S. M. & Langer, R. Wireless on-demand drug delivery. Nat. Electron. 4, 464–477 (2021).
Chen, J. et al. Enhancing aortic valve drug delivery with PAR2-targeting magnetic nano-cargoes for calcification alleviation. Nat. Commun. 15, 557 (2024).
Liu, Y.-L., Chen, D., Shang, P. & Yin, D.-C. A review of magnet systems for targeted drug delivery. J. Control. Rel. 302, 90–104 (2019).
Zhou, Z., Shen, Z. & Chen, X. Tale of two magnets: an advanced magnetic targeting system. ACS Nano 14, 7–11 (2020).
Carrey, J. & Hallali, N. Torque undergone by assemblies of single-domain magnetic nanoparticles submitted to a rotating magnetic field. Phys. Rev. B 94, 184420 (2016).
Leulmi, S. et al. Triggering the apoptosis of targeted human renal cancer cells by the vibration of anisotropic magnetic particles attached to the cell membrane. Nanoscale 7, 15904–15914 (2015).
Lee, J.-U. et al. Non-contact long-range magnetic stimulation of mechanosensitive ion channels in freely moving animals. Nat. Mater. 20, 1029–1036 (2021).
Wu, C. et al. Recent advances in magnetic-nanomaterial-based mechanotransduction for cell fate regulation. Adv. Mater. 30, 1705673 (2018).
Jang, J. T. et al. Critical enhancements of MRI contrast and hyperthermic effects by dopant-controlled magnetic nanoparticles. Angew. Chem. Int. Ed. 48, 1234–1238 (2009).
Guo, J., Yang, W. & Wang, C. Magnetic colloidal supraparticles: design, fabrication and biomedical applications. Adv. Mater. 25, 5196–5214 (2013).
Luo, B. et al. Mesoporous biocompatible and acid-degradable magnetic colloidal nanocrystal clusters with sustainable stability and high hydrophobic drug loading capacity. ACS Nano 5, 1428–1435 (2011).
Bai, F. et al. A versatile bottom-up assembly approach to colloidal spheres from nanocrystals. Angew. Chem. Int. Ed. 46, 6650–6653 (2007).
Kim, D. et al. Multiplexible wash-free immunoassay using colloidal assemblies of magnetic and photoluminescent nanoparticles. ACS Nano 11, 8448–8455 (2017).
Tay, A., Kunze, A., Murray, C. & Carlo, D. D. Induction of calcium influx in cortical neural networks by nanomagnetic forces. ACS Nano 10, 2331–2341 (2016).
Zhang, E. et al. Dynamic magnetic fields remote-control apoptosis via nanoparticle rotation. ACS Nano 8, 3192–3201 (2014).
Chen, M. et al. Remote control of mechanical forces via mitochondrial-targeted magnetic nanospinners for efficient cancer treatment. Small 16, 1905424 (2020).
Hillion, A. et al. Real-time observation and analysis of magnetomechanical actuation of magnetic nanoparticles in cells. Nano Lett. 22, 1986–1991 (2022).
Sebastian, V. in Magnetoelectric Polymer-Based Composites: Fundamentals and Applications (eds Lanceros-Méndez, S. & Martins, P.) 125–151 (Wiley, 2017).
Lee, J.-H. et al. Artificially engineered magnetic nanoparticles for ultra-sensitive molecular imaging. Nat. Med. 13, 95–99 (2007).
Lamouri, R. et al. Size effect on the magnetic properties of CoFe2O4 nanoparticles: a Monte Carlo study. Ceram. Int. 46, 8092–8096 (2020).
Kozielski, K. L. et al. Nonresonant powering of injectable nanoelectrodes enables wireless deep brain stimulation in freely moving mice. Sci. Adv. 7, eabc4189 (2021).
Zhang, Y. et al. Magnetoelectric nanoparticles incorporated biomimetic matrix for wireless electrical stimulation and nerve regeneration. Adv. Healthc. Mater. 10, 2100695 (2021).
Kim, Y. J. et al. Magnetoelectric nanodiscs enable wireless transgene-free neuromodulation. Nat. Nanotechnol. 20, 121–131 (2025).
Song, H. et al. Multi-target cell therapy using a magnetoelectric microscale biorobot for targeted delivery and selective differentiation of SH-SY5Y cells via magnetically driven cell stamping. Mater. Horiz. 9, 3031–3038 (2022).
Mushtag, F. et al. Magnetoelectric 3D scaffolds for enhanced bone cell proliferation. Appl. Mater. Today 16, 290–300 (2019).
Jang, J. & Park, C. B. Magnetoelectric dissociation of Alzheimer’s β-amyloid aggregates. Sci. Adv. 8, eabn1675 (2022).
Nair, M. et al. Externally controlled on-demand release of anti-HIV drug using magneto-electric nanoparticles as carriers. Nat. Commun. 4, 1707 (2013).
Rodzinski, A. et al. Targeted and controlled anticancer drug delivery and release with magnetoelectric nanoparticles. Sci. Rep. 6, 20867 (2016).
Zhang, C. et al. Synthesis of iron nanometallic glasses and their application in cancer therapy by a localized Fenton reaction. Angew. Chem. Int. Ed. 55, 2101–2106 (2016).
Yang, B., Chen, Y. & Shi, J. Reactive oxygen species (ROS)-based nanomedicine. Chem. Rev. 119, 4881–4985 (2019).
Lin, H., Chen, Y. & Shi, J. Nanoparticle-triggered in situ catalytic chemical reactions for tumour-specific therapy. Chem. Soc. Rev. 47, 1938–1958 (2018).
Zhou, Z., Song, J., Nie, L. & Chen, X. Reactive oxygen species generating systems meeting challenges of photodynamic cancer therapy. Chem. Soc. Rev. 45, 6597–6626 (2016).
Sies, H. et al. Defining roles of specific reactive oxygen species (ROS) in cell biology and physiology. Nat. Rev. Mol. Cell Biol. 23, 499–515 (2022).
Nath, I., Chakraborty, J. & Verpoort, F. Metal organic frameworks mimicking natural enzymes: a structural and functional analogy. Chem. Soc. Rev. 45, 4127–4170 (2016).
Zhang, R., Yan, X. & Fan, K. Nanozymes inspired by natural enzymes. Acc. Mater. Res. 2, 534–547 (2021).
Tang, Z., Zhao, P., Wang, H., Liu, Y. & Bu, W. Biomedicine meets Fenton chemistry. Chem. Rev. 121, 1981–2019 (2021).
Zhao, P., Li, H. & Bu, W. A forward vision for chemodynamic therapy: issues and opportunities. Angew. Chem. Int. Ed. 62, e202210415 (2023).
Lin, L.-S. et al. Synthesis of copper peroxide nanodots for H2O2 self-supplying chemodynamic therapy. J. Am. Chem. Soc. 141, 9937–9945 (2019).
Lin, L.-S. et al. Simultaneous Fenton-like ion delivery and glutathione depletion by MnO2-based nanoagent to enhance chemodynamic therapy. Angew. Chem. Int. Ed. 57, 4902–4906 (2018).
Zhou, Y., Fan, S., Feng, L., Huang, X. & Chen, X. Manipulating intratumoral Fenton chemistry for enhanced chemodynamic and chemodynamic-synergized multimodal therapy. Adv. Mater. 33, 2104223 (2021).
Tian, Q. et al. Recent advances in enhanced chemodynamic therapy strategies. Nano Today 39, 101162 (2021).
Tang, Z., Liu, Y., He, M. & Bu, W. Chemodynamic therapy: tumour microenvironment-mediated Fenton and Fenton-like reactions. Angew. Chem. Int. Ed. 58, 946–956 (2019).
Koo, S. et al. Enhanced chemodynamic therapy by Cu–Fe peroxide nanoparticles: tumor microenvironment-mediated synergistic Fenton reaction. ACS Nano 16, 2535–2545 (2022).
Feng, W. et al. Nanocatalysts-augmented and photothermal-enhanced tumor-specific sequential nanocatalytic therapy in both NIR-I and NIR-II biowindows. Adv. Mater. 31, 1805919 (2019).
Sang, Y. et al. Bioinspired construction of a nanozyme-based H2O2 homeostasis disruptor for intensive chemodynamic therapy. J. Am. Chem. Soc. 142, 5177–5183 (2020).
Chen, X. et al. NIR-triggered intracellular H+ transients for lamellipodia-collapsed antimetastasis and enhanced chemodynamic therapy. Angew. Chem. Int. Ed. 60, 21905–21910 (2021).
Shi, L. et al. An acidity-unlocked magnetic nanoplatform enables self-boosting ROS generation through upregulation of lactate for imaging-guided highly specific chemodynamic therapy. Angew. Chem. Int. Ed. 60, 9562–9572 (2021).
Liu, Y. et al. Engineering multifunctional RNAi nanomedicine to concurrently target cancer hallmarks for combinatorial therapy. Angew. Chem. Int. Ed. 57, 1510–1513 (2018).
Zhao, Z. et al. Reactive oxygen species-activatable liposomes regulating hypoxic tumor microenvironment for synergistic photo/chemodynamic therapies. Adv. Funct. Mater. 29, 1905013 (2019).
Sun, Q. et al. Recent advances on endogenous/exogenous stimuli-triggered nanoplatforms for enhanced chemodynamic therapy. Coord. Chem. Rev. 451, 214267 (2022).
Koo, S., Kim, Y. G., Lee, N., Hyeon, T. & Kim, D. Inorganic nanoparticle agents for enhanced chemodynamic therapy of tumors. Nanoscale 15, 13498–13514 (2023).
Kim, Y. G. et al. Ceria-based therapeutic antioxidants for biomedical applications. Adv. Mater. 36, 2210819 (2024).
Dutta, P. et al. Concentration of Ce3+ and oxygen vacancies in cerium oxide nanoparticles. Chem. Mater. 18, 5144–5146 (2006).
Nolan, M., Grigoleit, S., Sayle, D. C., Parker, S. C. & Watson, G. W. Density functional theory studies of the structure and electronic structure of pure and defective low index surfaces of ceria. Surf. Sci. 576, 217–229 (2005).
Esch, F. et al. Electron localization determines defect formation on ceria substrates. Science 309, 752–755 (2005).
Gupta, A., Das, S., Neal, C. J. & Seal, S. Controlling the surface chemistry of cerium oxide nanoparticles for biological applications. J. Mater. Chem. B 4, 3195–3202 (2016).
Soh, M. et al. Ceria–zirconia nanoparticles as an enhanced multi-antioxidant for sepsis treatment. Angew. Chem. Int. Ed. 56, 11399–11403 (2017).
Han, S. I. et al. Epitaxially strained CeO2/Mn3O4 nanocrystals as an enhanced antioxidant for radioprotection. Adv. Mater. 32, 2001566 (2020).
Jiang, P. et al. Tuning oxidant and antioxidant activities of ceria by anchoring copper single-site for antibacterial application. Nat. Commun. 15, 1010 (2024).
Chen, J., Patil, S., Seal, S. & McGinnis, J. F. Rare earth nanoparticles prevent retinal degeneration induced by intracellular peroxides. Nat. Nanotechnol. 1, 142–150 (2006).
He, L. et al. Highly bioactive zeolitic imidazolate framework-8-capped nanotherapeutics for efficient reversal of reperfusion-induced injury in ischemic stroke. Sci. Adv. 6, eaay9751 (2020).
Ji, W. et al. Self-catalytic small interfering RNA nanocarriers for synergistic treatment of neurodegenerative diseases. Adv. Mater. 34, 2105711 (2022).
Feng, Y. et al. A ferroptosis-targeting ceria anchored halloysite as orally drug delivery system for radiation colitis therapy. Nat. Commun. 14, 5083 (2023).
Fu, X. et al. Small molecule-assisted assembly of multifunctional ceria nanozymes for synergistic treatment of atherosclerosis. Nat. Commun. 13, 6528 (2022).
Im, G.-B. et al. Ceria nanoparticles as copper chaperones that activate SOD1 for synergistic antioxidant therapy to treat ischemic vascular diseases. Adv. Mater. 35, 2208989 (2023).
Yao, C. et al. Dynamic assembly of DNA–ceria nanocomplex in living cells generates artificial peroxisome. Nat. Commun. 13, 7739 (2022).
Weng, Q. et al. Catalytic activity tunable ceria nanoparticles prevent chemotherapy-induced acute kidney injury without interference with chemotherapeutics. Nat. Commun. 12, 1436 (2021).
Koo, S. et al. Ceria–vesicle nanohybrid therapeutic for modulation of innate and adaptive immunity in a collagen-induced arthritis model. Nat. Nanotechnol. 18, 1502–1514 (2023).
Lasting impact of lipid nanoparticles. Nat. Rev. Mater. 6, 1071 (2021).
van der Meel, R. et al. Smart cancer nanomedicine. Nat. Nanotechnol. 14, 1007–1017 (2019).
Shen, S. et al. A nanotherapeutic strategy to overcome chemotherapeutic resistance of cancer stem-like cells. Nat. Nanotechnol. 16, 104–113 (2021).
Li, Y. et al. Co-delivery of precisely prescribed multi-prodrug combination by an engineered nanocarrier enables efficient individualized cancer chemotherapy. Adv. Mater. 34, 2110490 (2022).
Janjua, T. I., Cao, Y., Yu, C. & Popat, A. Clinical translation of silica nanoparticles. Nat. Rev. Mater. 6, 1072–1074 (2021).
Zhang, Y. et al. Mesoporous carbon in biomedicine: modification strategies and biocompatibility. Carbon 212, 118121 (2023).
Wang, S. et al. Colloidal crystal engineering with metal-organic framework nanoparticles and DNA. Nat. Commun. 11, 2495 (2020).
Rabiee, N. Sustainable metal-organic frameworks (MOFs) for drug delivery systems. Mater. Today Commun. 35, 106244 (2023).
Huang, N., Wang, P. & Jiang, D. Covalent organic frameworks: a materials platform for structural and functional designs. Nat. Rev. Mater. 1, 16068 (2016).
Zhang, W. et al. Red-fluorescent covalent organic framework nanospheres for trackable anticancer drug delivery. ACS Appl. Mater. Interfaces 16, 342–352 (2024).
Geng, K. et al. Covalent organic frameworks: design, synthesis, and functions. Chem. Rev. 120, 8814–8933 (2020).
Burns, A., Ow, H. & Wiesner, U. Fluorescent core–shell silica nanoparticles: towards ‘Lab on a Particle’ architectures for nanobiotechnology. Chem. Soc. Rev. 35, 1028–1042 (2006).
Phillips, E. et al. Clinical translation of an ultrasmall inorganic optical-PET imaging nanoparticle probe. Sci. Transl Med. 6, 260ra149 (2014).
Gawne, P. J., Ferreira, M., Papaluca, M., Grimm, J. & Decuzzi, P. New opportunities and old challenges in the clinical translation of nanotheranostics. Nat. Rev. Mater. 8, 783–798 (2023).
Liu, J. et al. A sensitive and specific nanosensor for monitoring extracellular potassium levels in the brain. Nat. Nanotechnol. 15, 321–330 (2020).
Liu, J. et al. A highly sensitive and selective nanosensor for near-infrared potassium imaging. Sci. Adv. 6, eaax9757 (2020).
Yun, S. H. & Kwok, S. J. J. Light in diagnosis, therapy and surgery. Nat. Biomed. Eng. 1, 0008 (2017).
Chen, G., Qiu, H., Prasad, P. N. & Chen, X. Upconversion nanoparticles: design, nanochemistry, and applications in theranostics. Chem. Rev. 114, 5161–5214 (2014).
Montalti, M., Prodi, L., Rampazzo, E. & Zaccheroni, N. Dye-doped silica nanoparticles as luminescent organized systems for nanomedicine. Chem. Soc. Rev. 43, 4243–4268 (2014).
Li, Z. & Zhang, Y. Monodisperse silica-coated polyvinyl-pyrrolidone/NaYF4 nanocrystals with multicolor upconversion fluorescence emission. Angew. Chem. Int. Ed. 45, 7732–7735 (2006).
Liu, J. et al. Real-time in vivo quantitative monitoring of drug release by dual-mode magnetic resonance and upconverted luminescence imaging. Angew. Chem. Int. Ed. 53, 4551–4555 (2014).
Lee, J. E. et al. Uniform mesoporous dye-doped silica nanoparticles decorated with multiple magnetite nanocrystals for simultaneous enhanced magnetic resonance imaging, fluorescence imaging, and drug delivery. J. Am. Chem. Soc. 132, 552–557 (2010).
Li, Z., Barnes, J. C., Bosoy, A., Stoddart, J. F. & Zink, J. I. Mesoporous silica nanoparticles in biomedical applications. Chem. Soc. Rev. 41, 2590–2605 (2012).
Mora-Raimundo, P., Lozano, D., Manzano, M. & Vallet-Regi, M. Nanoparticles to knockdown osteoporosis-related gene and promote osteogenic marker expression for osteoporosis treatment. ACS Nano 13, 5451–5464 (2019).
Wang, X.-D., Rabe, K. S., Ahmed, I. & Niemeyer, C. M. Multifunctional silica nanoparticles for covalent immobilization of highly sensitive proteins. Adv. Mater. 27, 7945–7950 (2015).
Kim, M.-H. et al. Facile synthesis of monodispersed mesoporous silica nanoparticles with ultralarge pores and their application in gene delivery. ACS Nano 5, 3568–3576 (2011).
Hartono, S. B. et al. Poly-l-lysine functionalized large pore cubic mesostructured silica nanoparticles as biocompatible carriers for gene delivery. ACS Nano 6, 2104–2117 (2012).
Niu, D. et al. Monodispersed and ordered large-pore mesoporous silica nanospheres with tunable pore structure for magnetic functionalization and gene delivery. Adv. Mater. 26, 4947–4953 (2014).
Kwon, D. et al. Extra-large pore mesoporous silica nanoparticles for directing in vivo M2 macrophage polarization by delivering IL-4. Nano Lett. 17, 2747–2756 (2017).
Lee, B. et al. Co-delivery of metabolic modulators leads to simultaneous lactate metabolism inhibition and intracellular acidification for synergistic cancer therapy. Adv. Mater. 35, 2305512 (2023).
Ruskowitz, E. R. & DeForest, C. A. Photoresponsive biomaterials for targeted drug delivery and 4D cell culture. Nat. Rev. Mater. 3, 17087 (2018).
Yuan, Q. et al. Photon-manipulated drug release from a mesoporous nanocontainer controlled by azobenzene-modified nucleic acid. ACS Nano 6, 6337–6344 (2012).
Liu, J., Bu, W., Pan, L. & Shi, J. NIR-triggered anticancer drug delivery by upconverting nanoparticles with integrated azobenzene-modified mesoporous silica. Angew. Chem. Int. Ed. 52, 4375–4379 (2013).
Guardado-Alvarez, T. M., Devi, L. S., Russell, M. M., Schwartz, B. J. & Zink, J. I. Activation of snap-top capped mesoporous silica nanocontainers using two near-infrared photons. J. Am. Chem. Soc. 135, 14000–14003 (2013).
He, D., He, X., Wang, K., Cao, J. & Zhao, Y. A light-responsive reversible molecule-gated system using thymine-modified mesoporous silica nanoparticles. Langmuir 28, 4003–4008 (2012).
Mura, S., Nicolas, J. & Couvreur, P. Stimuli-responsive nanocarriers for drug delivery. Nat. Mater. 12, 991–1003 (2013).
Zhu, J. et al. Stimuli-responsive delivery vehicles based on mesoporous silica nanoparticles: recent advances and challenges. J. Mater. Chem. B 5, 1339–1352 (2017).
Wen, J. et al. Diverse gatekeepers for mesoporous silica nanoparticle based drug delivery systems. Chem. Soc. Rev. 46, 6024–6045 (2017).
Terstappen, G. C., Meyer, A. H., Bell, R. D. & Zhang, W. Strategies for delivering therapeutics across the blood–brain barrier. Nat. Rev. Drug Discov. 20, 362–383 (2021).
Tang, W. et al. Emerging blood–brain-barrier-crossing nanotechnology for brain cancer theranostics. Chem. Soc. Rev. 48, 2967–3014 (2019).
Baik, S.et al. Orally deliverable iron-ceria nanotablets for treatment of inflammatory bowel disease. Adv. Healthc. Mater. https://doi.org/10.1002/adhm.202401994 (2024).
Zhao, S. et al. An orally administered CeO2@montmorillonite nanozyme targets inflammation for inflammatory bowel disease therapy. Adv. Funct. Mater. 30, 2004692 (2020).
Naha, P. C. et al. Dextran-coated cerium oxide nanoparticles: a computed tomography contrast agent for imaging the gastrointestinal tract and inflammatory bowel disease. ACS Nano 14, 10187–10197 (2020).
Pearson, R. G. Hard and soft acids and bases. J. Am. Chem. Soc. 85, 3533–3539 (1963).
Kim, B. et al. Securing the payload, finding the cell, and avoiding the endosome: peptide-targeted, fusogenic porous silicon nanoparticles for delivery of siRNA. Adv. Mater. 31, 1902952 (2019).
Foss, D. V. et al. Peptide-mediated delivery of CRISPR enzymes for the efficient editing of primary human lymphocytes. Nat. Biomed. Eng. 7, 647–660 (2023).
Ngamcherdtrakul, W. et al. Cationic polymer modified mesoporous silica nanoparticles for targeted siRNA delivery to HER2+ breast cancer. Adv. Funct. Mater. 25, 2646–2659 (2015).
Shen, D. et al. Biphase stratification approach to three-dimensional dendritic biodegradable mesoporous silica nanospheres. Nano Lett. 14, 923–932 (2014).
Huang, P. et al. Metalloporphyrin-encapsulated biodegradable nanosystems for highly efficient magnetic resonance imaging-guided sonodynamic cancer therapy. J. Am. Chem. Soc. 139, 1275–1284 (2017).
Park, J.-H. et al. Biodegradable luminescent porous silicon nanoparticles for in vivo applications. Nat. Mater. 8, 331–336 (2009).
Du, B., Yu, M. & Zheng, J. Transport and interactions of nanoparticles in the kidneys. Nat. Rev. Mater. 3, 358–374 (2018).
Pan, L. et al. Cascade catalytic nanoparticles selectively alkalize cancerous lysosomes to suppress cancer progression and metastasis. Adv. Mater. 36, 2305394 (2024).
Kim, J. et al. Synergistic oxygen generation and reactive oxygen species scavenging by manganese ferrite/ceria co-decorated nanoparticles for rheumatoid arthritis treatment. ACS Nano 13, 3206–3217 (2019).
Kim, J. et al. Continuous O2-evolving MnFe2O4 nanoparticle-anchored mesoporous silica nanoparticles for efficient photodynamic therapy in hypoxic cancer. J. Am. Chem. Soc. 139, 10992–10995 (2017).
Zhou, Y. et al. Superexchange effects on oxygen reduction activity of edge-sharing [CoxMn1−xO6] octahedra in spinel oxide. Adv. Mater. 30, 1705407 (2018).
Li, J., Chu, D., Dong, H., Baker, D. R. & Jiang, R. Boosted oxygen evolution reactivity by igniting double exchange interaction in spinel oxides. J. Am. Chem. Soc. 142, 50–54 (2020).
Colombo, M. et al. Biological applications of magnetic nanoparticles. Chem. Soc. Rev. 41, 4306–4334 (2012).
Frey, N. A., Peng, S., Cheng, K. & Sun, S. Magnetic nanoparticles: synthesis, functionalization, and applications in bioimaging and magnetic energy storage. Chem. Soc. Rev. 38, 2532–2542 (2009).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT01770353 (2019).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT04682847 (2024).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT05359783 (2024).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT04261777 (2024).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT00368589 (2015).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT02319278 (2017).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT03948555 (2021).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT02511028 (2024).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT05357833 (2023).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT03619850 (2024).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT04268849 (2023).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT04278651 (2024).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT04205266 (2023).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT04080908 (2023).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT06271421 (2024).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT05010759 (2023).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT01266096 (2023).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT03465618 (2024).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT00848042 (2017).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT02680535 (2021).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT04240639 (2023).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT04656678 (2024).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT03667430 (2018).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT03823027 (2019).
Acknowledgements
This work was supported by the Institute for Basic Science (grant number IBS-R006-D1) and National Research Foundation of Korea (grant numbers 2022R1A2C1003527 and RS-2024-00350999).
Author information
Authors and Affiliations
Contributions
B.L., D.K. and T.H. contributed to the conception of the manuscript, writing the initial draft and designing the figures. All authors reviewed and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Materials thanks Jan Grimm, who co-reviewed with Karla Ximena Vazquez Prada; and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lee, B., Lee, Y., Lee, N. et al. Design of oxide nanoparticles for biomedical applications. Nat Rev Mater 10, 252–267 (2025). https://doi.org/10.1038/s41578-024-00767-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41578-024-00767-x